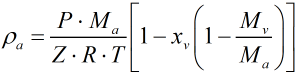C033 – Density of Moist Air
Description
This calculates the density of moist air from temperature, pressure, carbon dioxide content and one other parameter depending on the option selected.
References
Revised formula for the density of moist air (CIPM-2007) – A Picard, R S Davis, M Glaser and K Fujii
Kelton calculation reference C033
FLOCALC calculation reference F043
Options
Calculate from
- Relative Humidity
- Dew-point Temperature
- Vapour Mol Fraction
This option group allows the selection of the final parameter for calculating the density of moist air.
Constants
Molar gas constant – R = 8.314472 J mol-1K-1
Molar mass of water – Mv = 18.015 kg kmol-1
| Constituent | Molar mass, Mi (kg kmol-1) | Mole fraction, xi | Mi·xi |
|---|---|---|---|
| N2 | 28.0134 | 0.780848 | 21.874207 |
| O2 | 31.9988 | 0.209390 | 6.700229 |
| Ar | 39.948 | 0.009332 | 0.372795 |
| CO2 | 44.01 | 0.0004 | 0.017604 |
| Ne | 20.18 | 18.2 x 10-6 | 0.000367 |
| He | 4 | 5.2 x 10-6 | 21 x 10-6 |
| CH4 | 16 | 1.5 x 10-6 | 24 x 10-6 |
| Kr | 83.8 | 1.1 x 10-6 | 92 x 10-6 |
| H2 | 2 | 0.5 x 10-6 | 1 x 10-6 |
| N2O | 44 | 0.3 x 10-6 | 13 x 10-6 |
| CO | 28 | 0.2 x 10-6 | 6 x 10-6 |
| Xe | 131 | 0.1 x 10-6 | 13 x 10-6 |
| Σ | 28.96546 |
Calculation
Corrected Molar Mass of Air
The corrected molar mass of air is calculated from:
![]()
| Where | ||
|---|---|---|
| xCO2 | = | Carbon dioxide concentration |
Mole Fraction of Water Vapour
The mole fraction of water vapour is calculated from the formulae below. Use the appropriate one depending on the inputs available i.e. use equation on the far right if using dew point temperature as an input.
![]()
| Where | ||
|---|---|---|
| h | = | Relative humidity |
| P | = | Pressure |
| Psv | = | Saturated vapour pressure of moist air |
| f | = | Enhancement factor |
Saturated Vapour Pressure of Moist Air
The vapour pressure at saturation, Psv is calculated from:
![]()
| Where | ||
|---|---|---|
| T | = | Temperature in Kelvin |
| A | = | 1.2378847 x 10-5 K-2 |
| B | = | 1.9121316 x 10-2 K-1 |
| C | = | 33.93711047 |
| D | = | -6.3431645 x 103 K |
Enhancement Factor
The enhancement factor, f, is calculated from:
![]()
| Where | ||
|---|---|---|
| t | = | Temperature in °C |
| α | = | 1.0062 |
| β | = | 3.14 x 10-8 Pa -1 |
| γ | = | 5.6 x 10-7 K-2 |
Compressibility
The compressibility, Z, is calculated from:
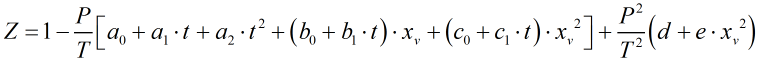
| Where | ||
| a0 | = | 1.58123 x 10-6 K Pa-1 |
| a1 | = | -2.9331 x 10-8 Pa-1 |
| a2 | = | 1.1043 x 10-10 K-1 Pa-1 |
| b0 | = | 5.707 x 10-6 K Pa-1 |
| b1 | = | -2.051 x 10-8 Pa-1 |
| c0 | = | 1.9898 x 10-4 K Pa-1 |
| c1 | = | -2.376 x 10-6 Pa-1 |
| d | = | 1.83 x 10-11 K2 Pa-2 |
| e | = | -0.765 x 10-8 K2 Pa-2 |
Density of Moist Air
The density of moist air, ρa is calculated from: